Dear readers,
Designing the world’s largest amateur rocket is not an easy task. It’s an iterative process with a lot of dependencies. One of the very important dependencies relates to the rocket’s « propellant delivery system », so how do you get fuel into the engine. There are several possibilities where a turbopump is clearly the sexiest, but also the hardest to master. On Nexø II, the fuel was delivered via the DPR system, which basically consisted of a tank of 20 liters of helium at 300 bar and a set of control valves. Let’s look at how such a system will look like in Spica size.
Let’s assume that Spica without fuel has a weight of 1700 kg and that its BPM100 engine is running at a chamber pressure of 15 bar. To bring this up to a height of 100 km, we need about 2000 kg fuel. At an OF ratio of 1.3, the LOX/ethanol has a medium density of approximately 1000 kg/m^3, so the size of the fuel tanks must be approximately 2000 liters. If we are to fly with 100% thrust all the way (we most likely won’t), then we have a volume of 2000 liters, which at MECO has to be pressurized to around 20 bars. It will require 40,000 standard liter (SL) of compressed gas (nitrogen) to achieve. It must of course be brought along in a compressed form, but it also has its price. During expansion from the high-pressure tank and down to 20 bar the gas is cooled and « loses » volume according to the ideal gas laws. The temperature drop is calculated easily as the final temperature is given as
T2 = T1 * (P2/P1)^((gamma-1)/gamma)
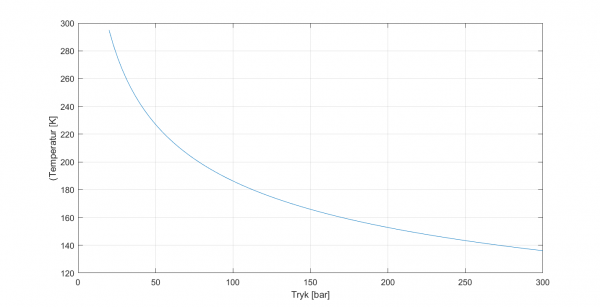
Where T1 is the starting temperature, P1 and P2 are start and end pressures, respectively, and gamma is the ratio of specific heats, which for nitrogen is 1.40. Below, I have plotted the end temperature as a function of starting pressure, assuming we expand to 20 bars starting from 295 K.
If we initially have 300 bars in the tank, then the expanded gas will have a final temperature of approx. 136 K, it will have « lost » more than half of its volume. Of course, it gets better the closer we get to P2, but overall, we lose almost 40% of the gas volume due to this cooling. If we need 40,000 SL, we must bring about 67,000 SL. If we use the same tank as for Nexø II, then we need 11 of these. They each weigh 12 kg, so we have gas tanks for a total of 132 kg. Then additionally we have tubes, fittings and valves …
It is therefore a heavy system. And an expensive system. The high-pressure tank on Nexø II was one of the most expensive single components of the rocket, with a price tag of around 10,000 kroner, imported from China. If anyone out there knows about large volume lightweight tanks at a reasonable price, we’ll be happy to hear from you.
Considering this, we’re looking at something else. We plan to introduce another cryogenic liquid on Spica. Besides, of course, having LOX on board, we are strongly considering taking the necessary nitrogen for pressurization in liquid form. If we bring only 40 liters of liquid nitrogen which you then evaporate and heat to 200-300 degrees, then you have all the gas you need.
Heat is available in ample amounts in the BPM100 engine, but we are a little nervous about having to implement a nitrogen heat exchanger in the engine, so we consider bringing a small burner that just has to evaporate the liquid nitrogen and heat it up. An important advantage of using a separate burner is the ability to test it without having to do a whole BPM100 test.
Nothing is certain at this point. We need to do some more calculations, but I find it very exciting. Though I’m not much for the extra complexity of having two cryogenic fluids on board. But in spaceflight, things rarely come without these kind of costs.






15 Comments
Comments are closed.